Ab initio DFT + U predictions of tensile properties of iron oxides
Abstract
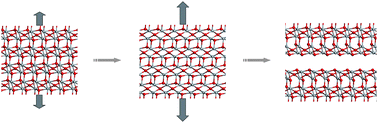
Stress-corrosion cracking of steel is a ubiquitous phenomenon in which steel is chemically corroded, followed by fracture induced by applied loads. The products of steel corrosion in moist air at high temperature (>570 °C) are α-Fe2O3, ![[1 with combining macron]](https://www.rsc.org/images/entities/char_0031_0304.gif) (3/
(3/

 Please wait while we load your content...
Please wait while we load your content...