Microfluidic electroporation of tumor and blood cells: observation of nucleus expansion and implications on selective analysis and purging of circulating tumor cells†
Abstract
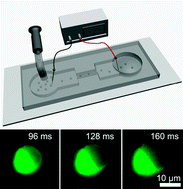
Circulating tumor
Maintenance work is planned for Wednesday 1st May 2024 from 9:00am to 11:00am (BST).
During this time, the performance of our website may be affected - searches may run slowly and some pages may be temporarily unavailable. If this happens, please try refreshing your web browser or try waiting two to three minutes before trying again.
We apologise for any inconvenience this might cause and thank you for your patience.
* Corresponding authors
a
Department of Agricultural and Biological Engineering, Purdue University, 225 S. University Street, West Lafayette, Indiana 47907, USA
E-mail:
changlu@purdue.edu
Fax: +1 765-496-1115
Tel: +1 765-494-1188
b Weldon School of Biomedical Engineering, Purdue University, 225 S. University Street, West Lafayette, Indiana 47907, USA
c School of Chemical Engineering, Purdue University, 225 S. University Street, West Lafayette, Indiana 47907, USA
d School of Public Health, Institute of Analytical Chemistry for Life Science, Nantong University, Nantong, China
Circulating tumor

 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?
 Fetching data from CrossRef.
Fetching data from CrossRef.
This may take some time to load.
Loading related content
