DFT study of the mechanism of benzocyclobutene formation by palladium-catalysed C(sp3)–H activation: role of the nature of the base and the phosphine†‡
Abstract
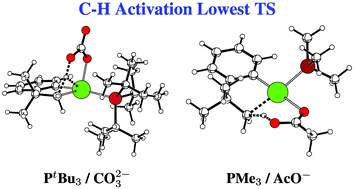
DFT(B3PW91) calculations of the mechanism of the intramolecular C(sp3)–H
- This article is part of the themed collection: Dalton Discussion 12: Catalytic C–H and C–X bond activation (DD12)

 Please wait while we load your content...
Please wait while we load your content...