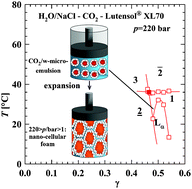
In this study we present a systematic investigation of the phase behaviour of microemulsions containing near- or supercritical solvents. The starting point of this study are microemulsions of the type water/NaCl–propane–polyethyleneglycol mono-n-alkyl ether at a pressure of p = 220 bar. Replacing propane stepwise by supercritical carbon dioxide the typical phase behavior of microemulsions systems can still be observed using scCO2 as the only nonpolar solvent. Thus, increasing the temperature a phase inversion from a CO2-in-water to a water-in-CO2 microemulsion via a balanced CO2 microemulsion is found for the first time. Such mixtures of water and scCO2 are expected to be versatile solvents in green chemistry. In addition, the formulation of supercritical microemulsions is the initial step in the PrincipleOfSupercriticalMicroemulsionExpansion (POSME) (DE Pat., 102 60 815 B4, 2008), which is a promising new approach for the production of low-cost nanocellular foams. In contrast to conventional foaming procedures, this approach suggests the formation of nanofoams by expanding micelles swollen with a supercritical blowing agent, thereby ensuring the unhindered formation and growth of bubbles without mass transport.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...