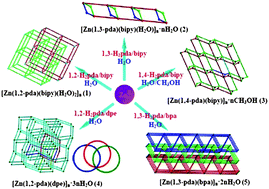
A series of new metal–organic frameworks (MOFs), [Zn(1,2-pda)(bipy)(H2O)2]n (1), [Zn(1,3-pda) (bipy)(H2O)]n·nH2O (2), [Zn(1,4-pda)(bipy)]n·nCH3OH (3), [Zn(1,2-pda)(dpe)]n·3nH2O (4), and [Zn(1,3-pda)(bpa)]n·2nH2O (5) [H2pda = phenylenediacetic acid, bipy = 4,4′-bipyridine, dpe = 1,2-di(4-pyridyl)ethylene, bpa = 1,2-bis(4-pyridyl)ethane] have been synthesized and structurally characterized. The structure determination reveals that complex 1 is a 2D layered network and exhibits a typical (4,4) topological net, which further assembles into a 3D two-fold interpenetrated pcu topology when hydrogen bonding interactions are considered. Complex 2 shows a 2D (5,2)-connected topological network via hydrogen bonding interactions based on the 1D double-chains. For complex 3, when the 1,4-pda ligands act as a three-connected node, the 2D double-layered structure displays a unique (5,3)-connected topology with a Schläfli symbol (42,67,8)(42,6). Complex 4 features a rare 3D three-fold interpenetrated diamondoid framework (dia, 66 topology) with (2.2.1) Hopf links and (6.3.3) Torus links, and the helical water chains are encased in the 1D neighboring channels. Complex 5 is a 2D corrugated net with a Schläfli symbol (44,62), and the puckered nature of the layers assembles them to interpenetrate in an unusual 2D → 3D parallel fashion. Particularly, a discrete tetramer water cluster, (H2O)4, is located in the crystal lattice of 5. The structural differences demonstrate that the backbones of the phenylenediacetic acids are a key point to form the final metal–ligand coordination polymers. Moreover, the fluorescent properties of complexes 1–5 were studied in the solid state at room temperature.

 Please wait while we load your content...
Please wait while we load your content...