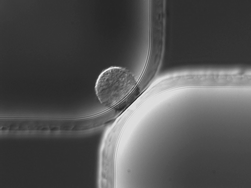
To enable several on-chip cell handling operations in a fused-silica substrate, small shallow micropores are radially embedded in larger deeper microchannels using an adaptation of single-level isotropic wet etching. By varying the distance between features on the photolithographic mask (mask distance), we can precisely control the overlap between two etch fronts and create a zero-thickness semi-elliptical micropore (e.g. 20 µm wide, 6 µm deep). Geometrical models derived from a hemispherical etch front show that micropore width and depth can be expressed as a function of mask distance and etch depth. These models are experimentally validated at different etch depths (25.03 and 29.78 µm) and for different configurations (point-to-point and point-to-edge). Good reproducibility confirms the validity of this approach to fabricate micropores with a desired size. To illustrate the wide range of cell handling operations enabled by micropores, we present three on-chip functionalities: continuous-flow particle concentration, immobilization of single cells, and picoliter droplet generation. (1) Using pressure differentials, particles are concentrated by removing the carrier fluid successively through a series of 44 shunts terminated by 31 µm wide, 5 µm deep micropores. Theoretical values for the concentration factor determined by a flow circuit model in conjunction with finite volume modeling are experimentally validated. (2) Flowing macrophages are individually trapped in 20 µm wide, 6 µm deep micropores by hydrodynamic confinement. The translocation of transcription factor NF-κB into the nucleus upon lipopolysaccharide stimulation is imaged by fluorescence microscopy. (3) Picoliter-sized droplets are generated at a 20 µm wide, 7 µm deep micropore T-junction in an oil stream for the encapsulation of individual E. coli bacteria cells.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...