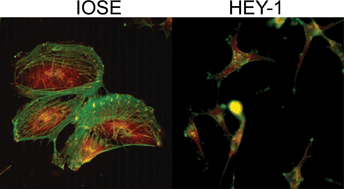
Ovarian cancer is the deadliest gynecological cancer, which may arise in part due to the concurrent invasion and metastasis of high grade tumors. It is thus crucial to gain insight into the adhesion and migration mechanisms in vivo, as this may ultimately lead to new treatment/detection options. To explore this possibility, we have used multiphoton excited photochemistry (MPE) to synthesize models of the ovarian basal lamina consisting of crosslinked laminin nanofibers to quantify the adhesion/migration dynamics. The nanostructured laminin patterns permit the systematic comparison of total migration, directed migration, adhesion, and morphology of “normal” immortalized human ovarian epithelial cells (IOSE) and three lines of varying metastatic potential (OVCA433, SKOV-3.ip1, and HEY-1 cells). We find that the migration of all the cell lines is directed by the crosslinked fibers, and that the contact guidance enhances the total migration rates relative to monolayers. These rates increase with increasing metastatic potential, and the more invasive cells are less rigid and more weakly adhered to the nanofibers. The extent of directed migration also depends on the cell polarity and focal adhesion expression. For the invasive cells, these findings are similar to the integrin-independent ameboid-like migration seen for polar cells in collagen gels. Collectively, the results suggest that contact mediated migration as well as decreased adhesion may be operative in metastasis of ovarian cancer in vivo.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...