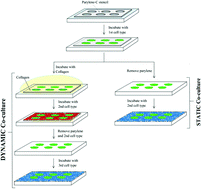
Many biological processes, such as stem cell differentiation, wound healing and development, involve dynamic interactions between cells and their microenvironment. The ability to control these dynamic processes in vitro would be potentially useful to fabricate tissue engineering constructs, study biological processes, and direct stem cell differentiation. In this paper, we used a parylene-C microstencil to develop two methods of creating patterned co-cultures using either static or dynamic conditions. In the static case, embryonic stem (ES) cells were co-cultured with fibroblasts or hepatocytes by using the reversible sealing of the stencil on the substrate. In the dynamic case, ES cells were co-cultured with NIH-3T3 fibroblasts and AML12 hepatocytes sequentially by engineering the surface properties of the stencil. In this approach, the top surface of the parylene-C stencil was initially treated with hyaluronic acid (HA) to reduce non-specific cell adhesion. The stencil was then sealed on a substrate and seeded with ES cells which adhered to the underlying substrate through the holes in the membrane. To switch the surface properties of the parylene-C stencils to cell adhesive, collagen was deposited on the parylene-C surfaces. Subsequently, a second cell type was seeded on the parylene-C stencils to form a patterned co-culture. This group of cells was removed by peeling off the parylene-C stencils, which enabled the patterning of a third cell type. Although the static patterned co-culture approach has been demonstrated previously with a variety of methods, layer-by-layer modification of microfabricated parylene-C stencils enables dynamic patterning of multiple cell types in sequence. Thus, this method is a promising approach to engineering the complexity of cell–cell interactions in tissue culture in a spatially and temporally regulated manner.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...