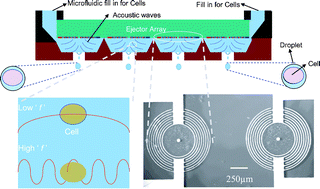
The capability to encapsulate single to few cells with micrometre precision, high viability, and controlled directionality via a nozzleless ejection technology using a gentle acoustic field would have great impact on tissue engineering, high throughput screening, and clinical diagnostics. We demonstrate encapsulation of single cells (or a few cells) ejected from an open pool in acoustic picolitre droplets. We have developed this technology for the specific purpose of printing cells in various biological fluids, including PBS and agarose hydrogels used in tissue engineering. We ejected various cell types, including mouse embryonic stem cells, fibroblasts, AML-12 hepatocytes, human Raji cells, and HL-1 cardiomyocytes encapsulated in acoustic picolitre droplets of around 37 µm in diameter at rates varying from 1 to 10 000 droplets per second. At such high throughput levels, we demonstrated cell viabilities of over 89.8% across various cell types. Moreover, this ejection method is readily adaptable to other biological applications, such as extracting data from single cells and generating large cell populations from single cells. The technique described in the current study may also be applied to investigate stem cell differentiation at the single cell level, to direct tissue printing, and to isolating pure RNA or DNA from a single cell at the picolitre level. Overall, the techniques described have the potential for widespread impact on many high-throughput testing applications in the biological and health sciences.

 Please wait while we load your content...
Please wait while we load your content...