A parametric study of human fibroblasts culture in a microchannel bioreactor
Abstract
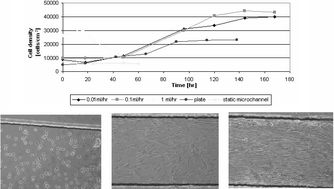
The culture of cells in a microbioreactor can be highly beneficial for cell biology studies and tissue engineering applications. The present work provides new insights into the relationship between cell growth, cell morphology, perfusion rate, and design parameters in microchannel bioreactors. We demonstrate the long-term culture of mammalian (human foreskin fibroblasts, HFF) cells in a microbioreactor under constant perfusion in a straightforward simple manner. A perfusion system was used to culture human cells for more than two weeks in a plain microchannel (130 µm × 1 mm × 2 cm). At static conditions and at high flow rates (>0.3 ml h−1), the cells did not grow in the microchannel for more than a few days. For low flow rates (<0.2 ml h−1), the cells grew well and a confluent layer was obtained. We show that the culture of cells in microchannels under perfusion, even at low rates, affects cell growth kinetics as well as cell morphology. The oxygen level in the microchannel was evaluated using a mass transport model and the maximum cell density measured in the microchannel at steady state. The maximum shear stress, which corresponds to the maximum flow rate used for long term culture, was 20 mPa, which is significantly lower than the shear stress cells may endure under physiological conditions. The effect of channel size and cell type on long term cell culture were also examined and were found to be significant. The presented results demonstrate the importance of understanding the relationship between design parameters and cell behavior in microscale culture system, which vary from physiological and traditional culture conditions.

 Please wait while we load your content...
Please wait while we load your content...