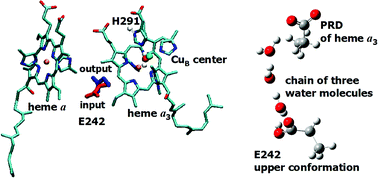
Cytochrome c oxidase (CcO) is the terminal enzyme in the respiratory electron transport chain of aerobic organisms. It catalyses the reduction of atmospheric oxygen to water, and couples this reaction to proton pumping across the membrane; this process generates the electrochemical gradient that subsequently drives the synthesis of ATP. The molecular details of the mechanism by which electron transfer is coupled to proton pumping in CcO is poorly understood. Recent calculations from our group indicate that His291, a ligand of the CuB center of the enzyme, may play the role of the pumping element. In this paper we describe calculations in which a DFT/continuum electrostatic method is used to explore the coupling of the conformational changes of Glu242 residue, the main proton donor of both chemical and pump protons, to its pKa, and the pKa of His291, a putative proton loading site of our pumping model. The computations are done for several redox states of metal centers, different protonation states of Glu242 and His291, and two well-defined conformations of the Glu242 side chain. Thus, in addition to equilibrium redox/protonation states of the catalytic cycle, we also examine the transient and intermediate states. Different dielectric models are employed to investigate the robustness of the results, and their viability in the light of the proposed proton pumping mechanism of CcO. The main results are in agreement with the experimental measurements and support the proposed pumping mechanism. Additionally, the present calculations indicate a possibility of gating through conformational changes of Glu242; namely, in the pumping step, we find that Glu242 needs to be reprotonated before His291 can eject a proton to the P-site of membrane. As a result, the reprotonation of Glu can control proton release from the proton loading site.

 Please wait while we load your content...
Please wait while we load your content...