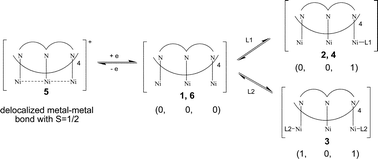
This study provides an opportunity to control the magnetic spin of nickel atoms using various mixed anionic ligands. A series of linear trinickel complexes supported by two kinds of ligands, oligo-α-pyridylamido and sulfonyl amido/amido, were synthesized and their structures were determined by X-ray diffraction. The three nickel atoms of [Ni3(Lpts)2(dpa)2] (1) (dpa− = dipyridylamido, Lpts2− = N,N′-bis(p-toluenesulfonyl)pyridyldiamido) display short Ni–N (∼1.90 Å) bond distances, which are consistent with a low spin state of Ni(II) ions, and exhibit spin states of (0, 0, 0) for the three Ni(II) ions. One of the terminal Ni(II) ions of [Ni3(Lms)2(dpa)2(H2O)] (2) (Lms2− = N,N′-bis(4-methylsulfonyl)-pyridyldiamido) and [Ni3(Lpts)2(pepteaH2)] (4) (pepteaH22− = pentapyridyldiamidodiamine) bonded with an axial ligand exhibits a square pyramidal (NiN4X) geometry with long Ni–N bond distances (∼2.10 Å) which are consistent with a high spin Ni(II) configuration. The spin states of these trinickel complexes are (1, 0, 0). Complex 2 and 6 can be interchanged by the removal or addition of an axial water molecule. The structural features of 6 are comparable with those of 1. Both the terminal Ni(II) ions in [Ni3(LAc)2(dpa)2] (3) (Lac2− = N,N′-biacetyl-pyridyldiamido) are in square pyramidal geometry and exhibit high spin. The spin states of the nickel ions in 3 are (1, 0, 1), and the two terminal nickel ions exhibit antiferromagnetic interactions. The molecular structure of [Ni3(Lpts)2(dpa)2](BF4) (5), which was obtained by the one-electron oxidation of 1, is similar to those of the neutral analogue 1, except for the presence of a counter anion to compensate for the positive charge on the Ni3 core. All of the Ni–Ni bond lengths of 5 are slightly shorter (ca. 0.05 Å) than those in the neutral analogues. This is attributed to the formation of partial Ni–Ni bonding.

 Please wait while we load your content...
Please wait while we load your content...