Hypercarbons in polyhedral structures
Abstract
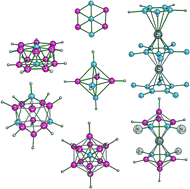
Though carbon is mostly tetravalent and tetracoordinated, there are several examples where the coordination number exceeds four. Structural varieties that exhibit hypercarbons in polyhedral structures such as polyhedral carboranes, sandwich complexes, encapsulated polyhedral structures and novel planar aromatic systems with atoms embedded in the middle are reviewed here. The structural variety anticipated with hypercoordinate carbon among carboranes is large as there are many modes of condensation that could lead to large number of new patterns. The relative stabilities of positional isomers of polyhedral carboranes, sandwich structures, and endohedral carboranes are briefly described. The mno rule accounts for the variety of structural patterns. Wheel-shaped and planar hypercoordinated molecules are recent theoretical developments in this area.

 Please wait while we load your content...
Please wait while we load your content...