The Walden cycle revisited: a computational study of competitive ring closure to α- and β-lactones
Abstract
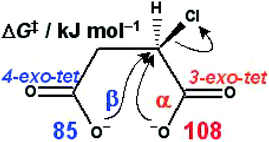
The text-book Walden cycle which interconverts the stereochemical configurations of chlorosuccinic and malic acids involves a β-lactone intermediate in preference to an α-lactone intermediate because the Onuc C Cl angle in the transition structure for the former (174°) is more favourable than that for the latter (139°), as determined by PCM(ε = 78.4)/B3LYP/6-31+G* calculations; the smaller ring-strain energy of the β-lactone contributes little to the reactivity difference.

 Please wait while we load your content...
Please wait while we load your content...