Water, sulfur dioxide and nitric acid adsorption on calcium carbonate: A transmission and ATR-FTIR study
Abstract
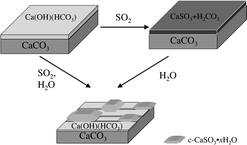
Calcium carbonate (CaCO3) is a reactive component of mineral dust aerosol as well as buildings, statues and monuments. In this study, attenuated total reflection (ATR) and transmission Fourier transform infrared spectroscopy (FTIR) have been used to study the uptake of water, sulfur dioxide and nitric acid on CaCO3 particles at 296 K. Under atmospheric conditions, CaCO3 particles are terminated by a Ca(OH)(CO3H) surface layer. In the presence of water vapor between 5 and 95% relative humidity (RH), water molecularly adsorbs on the Ca(OH)(CO3H) surface resulting in the formation of an adsorbed thin water film. The adsorbed water film assists in the enhanced uptake of sulfur dioxide and nitric acid on CaCO3 in several ways. Under dry conditions (near 0% RH), sulfur dioxide and nitric acid react with the Ca(OH)(CO3H) surface to form adsorbed carbonic acid (H2CO3) along with sulfite and nitrate, respectively. Adsorbed carbonic acid is stable on the surface under vacuum conditions. Once the surface saturates with a carbonic acid capping layer, there is no additional uptake of gas-phase sulfur dioxide and nitric acid. However, upon adsorption of water, carbonic acid dissociates to form gaseous carbon dioxide and there is further uptake of sulfur dioxide and nitric acid. In addition, adsorbed water increases the mobility of the ions at the surface and enhances uptake of SO2 and HNO3. In the presence of adsorbed water, CaSO3 forms islands of a crystalline hydrate whereas Ca(NO3)2 forms a deliquescent layer or micropuddles. Thus adsorbed water plays an important and multi-faceted role in the uptake of pollutant gases on CaCO3.

 Please wait while we load your content...
Please wait while we load your content...