Catalytic asymmetric synthesis of enantiopure isoprenoid building blocks: application in the synthesis of apple leafminer pheromones†
Abstract
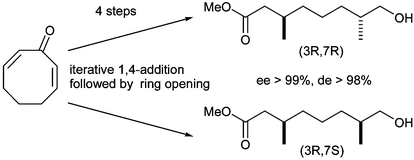
The first catalytic asymmetric procedure capable of preparing all 4 diastereoisomers (ee > 99%, de > 98%) of a versatile saturated

 Please wait while we load your content...
Please wait while we load your content...