o-(Cyclohexyliminoethyl)(arylamido)benzene and related complexes of zirconium and titanium. The non-innocence of the ketimino functionality
Abstract
N-Arylamido complexes of zirconium in which the amido functional group is attached to an o-(alkyliminoethyl) substituted aromatic ring, have been synthesised by salt elimination reactions and characterised by spectroscopic and diffraction methods; they are analogous to the N-silylamido species recently reported (Dalton Trans., 2002, 3290–3299). The ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(CH3)]C6H4N(H)(xyl), LxylH, and 2-[CyN
C(CH3)]C6H4N(H)(xyl), LxylH, and 2-[CyN![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(CH3)]C6H4N(H)(mes) LmesH, Cy = C6H11, xyl = 3,5-Me2C6H3 mes = 2,4,6-Me3C6H2, were prepared in good yields by Buchwald–Hartwig
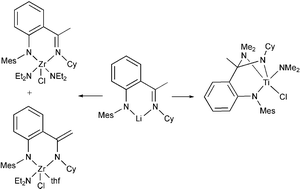
C(CH3)]C6H4N(H)(mes) LmesH, Cy = C6H11, xyl = 3,5-Me2C6H3 mes = 2,4,6-Me3C6H2, were prepared in good yields by Buchwald–Hartwig ![[double bond, length as m-dash]](https://www.rsc.org/images/entities/char_e001.gif) C(CH3)]C6H4NH2. Reaction of LmesLi with Zr(NEt2)2Cl2(thf)2 gave after chloride substitution the arylamido ketimino complex LmesZr(NEt2)2Cl 1; variable amounts of the arylamido vinylamido complex 2 were also obtained. Interaction of LmesLi or LxylLi with Ti(NMe2)2Cl2 gave rise to the tripodal bis-amido amino complexes 5 and 6 possibly formed by
C(CH3)]C6H4NH2. Reaction of LmesLi with Zr(NEt2)2Cl2(thf)2 gave after chloride substitution the arylamido ketimino complex LmesZr(NEt2)2Cl 1; variable amounts of the arylamido vinylamido complex 2 were also obtained. Interaction of LmesLi or LxylLi with Ti(NMe2)2Cl2 gave rise to the tripodal bis-amido amino complexes 5 and 6 possibly formed by

 Please wait while we load your content...
Please wait while we load your content...