Silver(i) oxide–silver halide mediated alcoholysis of O-benzoyl-myo-inositol 1,3,5-orthoformates: intramolecular assistance by the sulfonyl group
Abstract
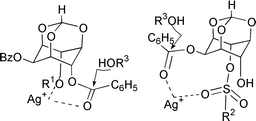
Silver(I) oxide–silver halide mediated alcoholyses of racemic 2,4-di-O-benzoyl-myo-inositol 1,3,5-orthoformate, and its 6-O-methyl and 6-O-sulfonylated derivatives, under identical conditions have been compared. While only the 4-O-benzoyl group undergoes solvolysis in the former two, to yield the corresponding 2-O-benzoyl-myo-inositol 1,3,5-orthoformate, both the 4-O- as well as the 2-O-benzoyl groups undergo solvolysis in the latter, to yield racemic 6-O-sulfonyl-myo-inositol 1,3,5-orthoformates. These results show that solvolysis of the 2-O-benzoyl group in sulfonates is a consequence of intramolecular assistance by the sulfonyl group. Catalytic efficiency of the silver halides in bringing about solvolysis of the benzoates decreased in the order AgI > AgBr > AgCl. A reaction mechanism involving silver–inositol derivative chelates has been proposed.

 Please wait while we load your content...
Please wait while we load your content...