Following the structural changes in iron phosphate catalysts by in situ combined XRD/QuEXAFS technique
Abstract
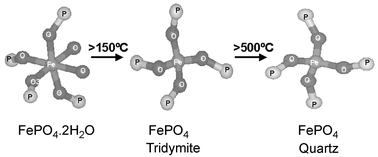
The structure of iron phosphate catalysts prepared by three different synthetic methods has been studied using in situ combined XRD/QuEXAFS. Two of the synthesis procedures have previously been employed to produce these catalysts whilst the third is a new method employing hydrothermal techniques. The combined XRD/QuEXAFS approach supported by ex situ measurements carried out using XRD, TGA and surface area measurements, showed that all three methods produced a catalyst precursor material, having the chemical composition of FePO4·2H2O. The in situ study clearly shows the loss of two water molecules during the initial stages of heating and a transformation to FePO4 having tridymite type structure takes place at temperatures above ca. 100 °C. The crystallinity of the tridymite phase appeared to improve upon heating to ca. 500 °C and above which a further transformation to a quartz phase takes place. Of the three catalysts, the material produced by hydrothermal methods was found to be the most phase pure and to possess a higher surface area than the materials prepared by the other two methods.

 Please wait while we load your content...
Please wait while we load your content...