Study on the structural and electrical properties of the double perovskite oxide SrMn0.5Nb0.5O3−δ
Abstract
The new non-stoichiometric mixed perovskite SrMn0.5Nb0.5O3−δ has a cubic double perovskite structure similar to that of Sr2CrNbO6 with space group Fm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m
(225), a
= 7.9338(3)
Å, V
= 499.39(6)
Å3 according to X-ray diffraction. The material is redox stable and maintains its structure in a reducing atmosphere. After reducing in 5% H2 at 900 °C for 6 hours, SrMn0.5Nb0.5O3−δ still exhibits a cubic structure with space group Pm
m
(225), a
= 7.9338(3)
Å, V
= 499.39(6)
Å3 according to X-ray diffraction. The material is redox stable and maintains its structure in a reducing atmosphere. After reducing in 5% H2 at 900 °C for 6 hours, SrMn0.5Nb0.5O3−δ still exhibits a cubic structure with space group Pm![[3 with combining macron]](https://www.rsc.org/images/entities/char_0033_0304.gif) m
(221), a
= 4.0022(5)
Å, V
= 64.10(8)
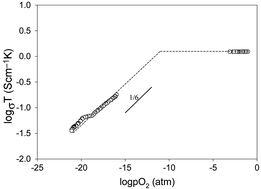
Å3. A lattice volume expansion of 2.7% was observed during the reduction. TGA analysis indicates SrMn0.5Nb0.5O3−δ
loses 0.125 oxygen per formula unit from 500 to 950 °C in 5% H2. This weight change is consistent with a
m
(221), a
= 4.0022(5)
Å, V
= 64.10(8)
Å3. A lattice volume expansion of 2.7% was observed during the reduction. TGA analysis indicates SrMn0.5Nb0.5O3−δ
loses 0.125 oxygen per formula unit from 500 to 950 °C in 5% H2. This weight change is consistent with a

 Please wait while we load your content...
Please wait while we load your content...