Novel polynuclear CuII/CoII complexes constructed from one and two Cu2Co triangles with antiferromagnetic exchange coupling
Abstract
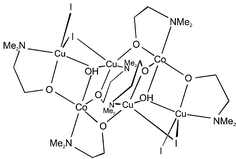
The reaction of acetonitrile solutions of CoX2·2H2O (X = NCS, I, Br) and 2-dimethylaminoethanol (HL) with copper powder in air leads to the formation of new tri- and hexa-nuclear complexes [Cu2Co(NCS)3L3]·½CH3CN (1), [Cu2Co(μ3-OH)I2L3]2·4CH3CN (2) and [Cu2Co(μ3-OH)Br2L3]2·2CH3CN (3). X-Ray crystallographic analysis of 1 reveals a symmetrical triangular core with one Co and two Cu atoms. The amino alkoxo- and SNCS-bridges between metal centres result in copper–copper and copper–cobalt separations of 3.413(2) and 3.103(2) Å, respectively. Additional intermolecular association occurs through the NCS groups bridging Co and Cu centres of adjacent complex molecules to give chains. In the solid-state structures of 2 and 3 two symmetry-related Cu2Co units are linked by amino alkoxo bridges to form a hexanuclear molecule with intermetallic distances ranging from 3.102(2)–3.260(2) (Cu⋯Co) to 3.287(3)–3.931(2) Å (Cu⋯Cu). The triangular planes formed by two copper and one cobalt atoms bridged by oxygen atoms from L groups are capped by a μ3-hydroxide not found in the core of 1. The hexanuclear units show no significant intermolecular contacts in the solid state. Variable-temperature magnetic susceptibility studies performed on 1 and 2 in the temperature range 5–280 K gave satisfactory fits to the observed susceptibility data by assuming isotropic magnetic exchange interactions and using the appropriate spin Hamiltonians considering 1 as a trinuclear entity and 2 as a combination of two trinuclear entities. An antiferromagnetic spin exchange operates in the Cu2Co units of both complexes [JCuCu = 40.1(4) (1), 48.7(9) (2); JCuCo = 141(2) (1), 86.2(1.3) cm−1 (2)] while a weak ferromagnetic coupling is active between Cu and Co ions from different trinuclear entities in 2 (JCuCo′ = −8.0(4) cm−1).

 Please wait while we load your content...
Please wait while we load your content...