Sonication induced sheet formation at the air–water interface†
Abstract
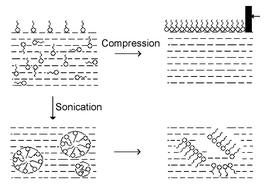
A hydrophobic pentadecapeptide, AGAAAAGAVVGGLGG (1), part of the prion sequence PrP (106-127), on fresh aqueous dissolution takes a mixture of random and sheet conformations which forms a stable monolayer with a high β-sheet content when compressed at the air–water interface. This also develops into a kinetically stabilized β-sheet structure on

 Please wait while we load your content...
Please wait while we load your content...