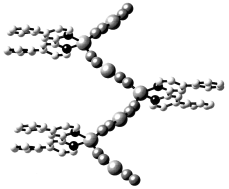
The structural consequences of introducing sterically demanding substituents have been investigated for composite organic–inorganic materials of the copper cyanide–organodiimine system. The hydrothermal reactions of copper(I) cyanide and KCN, with the appropriate chelating organodiimine ligands yield a series of one-dimensional copper cyanide chains decorated either with linear {(LL)Cu(CN)} (LL = organodiimine) side chains or with diimine ligands bound directly to the copper sites of the parent chain. The structures of [Cu3(CN)3(2,2′-bpy)] (1) (2,2′-bpy = 2,2′-bipyridine) and [Cu3(CN)3(phen)] (2) (phen = 1,10-phenanthroline) are similar to that of the previously reported [Cu4(CN)4(biquin)] (biquin = 2,2′-biquinoline),
exhibiting the characteristic one-dimensional {Cu(CN)} chain with diimine–copper cyanide side arms. In contrast, the structures of [Cu3(CN)3(biquin)2] (3), [Cu2(CN)2(dpbp)], (4) (dpbp = 4,4′-diphenyl-2,2′-bipyridine), and [Cu(CN)(dpphen)] (5) (dpphen = 4,7-diphenyl-1,10-phenanthroline) are characterized by direct attachment of the organodiimine ligand to the copper cyanide chain.
You have access to this article
 Please wait while we load your content...
Something went wrong. Try again?
Please wait while we load your content...
Something went wrong. Try again?


 Please wait while we load your content...
Please wait while we load your content...