Stereodivergent, Diels–Alder-initiated organocascades employing α,β-unsaturated acylammonium salts: scope, mechanism, and application†
Abstract
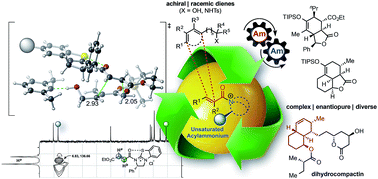
Chiral α,β-unsaturated acylammonium salts are novel dienophiles enabling enantioselective Diels–Alder-lactonization (DAL) organocascades leading to cis- and trans-fused, bicyclic γ- and δ-lactones from readily prepared dienes, commodity acid chlorides, and a chiral isothiourea organocatalyst under mild conditions. We describe extensions of stereodivergent DAL organocascades to other racemic dienes bearing pendant secondary and tertiary alcohols, and application to a formal synthesis of (+)-dihydrocompactin is described. A combined experimental and computational investigation of unsaturated acylammonium salt formation and the entire DAL organocascade pathway provide a rationalization of the effect of Brønsted base additives and enabled a controllable, diastereodivergent DAL process leading to a full complement of possible stereoisomeric products. Evaluation of free energy and enthalpy barriers in conjunction with experimentally observed temperature effects revealed that the DAL is a rare case of an entropy-controlled diastereoselective process. NMR analysis of diene alcohol–Brønsted base interactions and computational studies provide a plausible explanation of observed stabilization of exo transition-state structures through hydrogen-bonding effects.


 Please wait while we load your content...
Please wait while we load your content...