Insights into water-mediated ion clustering in aqueous CaSO4 solutions: pre-nucleation cluster characteristics studied by ab initio calculations and molecular dynamics simulations†
Abstract
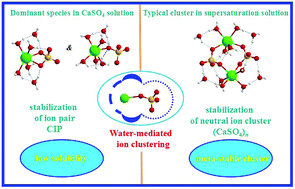
The molecular structure of growth units building crystals is a fundamental issue in the crystallization processes from aqueous solutions. In this work, a systematic investigation of pre-nucleation clusters and their hydration characteristics in aqueous CaSO4 solutions was performed using ab initio calculations and molecular dynamics (MD) simulations. The results of ab initio calculations and MD simulations indicate that the dominant species in aqueous CaSO4 solutions are monodentate ion-associated structures. Compared with charged ion clusters, neutral clusters are more likely to be present in an aqueous CaSO4 solution. Neutral (CaSO4)m clusters are probably the growth units involved in the pre-nucleation or crystallization processes. Meanwhile, hydration behavior around ion associated species in aqueous CaSO4 solutions plays an important role in related phase/polymorphism selections. Upon ion clustering, the residence of some water molecules around Ca2+ in ion-associated species is weakened while that of some bridging waters is enhanced due to dual interaction by Ca2+ and SO42−. Some phase/polymorphism selections can be achieved in aqueous CaSO4 solutions by controlling the hydration around pre-nucleation clusters. Moreover, the association trend between calcium and sulfate is found to be relatively strong, which hints at the low solubility of calcium sulfate in water.


 Please wait while we load your content...
Please wait while we load your content...